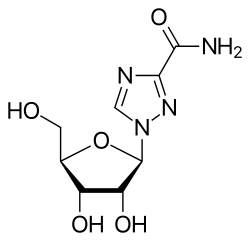
利巴韦林
此條目可参照英語維基百科相應條目来扩充。 |
 | |
 | |
| 臨床資料 | |
|---|---|
| 读音 | /ˌraɪbəˈvaɪrɪn/ RY-bə-VY-rin |
| 商品名 | Copegus、Rebetol、Virazole及其他[1] |
| 其他名稱 | 1-(β-D-Ribofuranosyl)-1"H"-1,2,4-triazole-3-carboxamide, tribavirin (BAN UK) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605018 |
| 核准狀況 | |
| 懷孕分級 |
|
| 给药途径 | 口服給藥, 肺部給藥 |
| ATC碼 | |
| 法律規範狀態 | |
| 法律規範 |
|
| 藥物動力學數據 | |
| 生物利用度 | 64%[3] |
| 血漿蛋白結合率 | 0%[3] |
| 药物代谢 | 肝臟及細胞內[3] |
| 生物半衰期 | 298小時 (多次劑量),43.6小時 (單次劑量)[3] |
| 排泄途徑 | 尿液 (61%), 糞便 (12%)[3] |
| 识别信息 | |
| |
| CAS号 | 36791-04-5 |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| NIAID ChemDB | |
| PDB配體ID | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.164.587 |
| 化学信息 | |
| 化学式 | C8H12N4O5 |
| 摩尔质量 | 244.21 g·mol−1 |
| 3D模型(JSmol) | |
| 熔点 | 166至168 °C(331至334 °F) |
| |
| |
利巴韋林(INN:ribavirin)以品牌名稱Virazole(俗稱病毒唑)等於市場上銷售,是一種抗病毒藥物,用於治療人類呼吸道合胞病毒(RSV)、C型肝炎病毒(HCV)感染以及某些病毒性出血熱。[1]針對C型肝炎,它會與其他藥物併用,例如司美匹韋、索磷布韋、聚乙二醇化干擾素α-2b或聚乙二醇化干擾素α-2a。[1]它也可用於治療病毒性出血熱-特別是拉薩熱、克里米亞-剛果出血熱以及漢他病毒感染(但伊波拉出血熱或馬堡病毒出血熱除外)。[1]雖然利巴韋林在臨床上已應用多年,但進入21世紀後,其地位因缺乏實證醫學支持而受到挑戰。針對過去常見的某些病毒感染適應症,目前的臨床研究並未證實其具備預期療效,因此引發醫學界對其療效與安全性的重新評估。[5][6]
使用利巴韋林常見的副作用有疲勞、頭痛、噁心、發燒、肌肉疼痛以及情緒易怒。[1]嚴重的副作用則有紅血球分解(溶血反應)、肝臟問題以及過敏反應 。[1]孕婦使用此藥物可能會對發育中的胎兒造成傷害。[1]因此建議男性與女性使用者在用藥期間及停藥後至少七個月內,皆須採取有效的避孕措施。[7]利巴韋林的作用機制目前尚未被完全了解。[1]
利巴韋林於1971年取得專利,並於1986年獲准用於醫療用途 。[8]它已收錄在世界衛生組織基本藥物標準清單之中。[9]目前市面上已有該藥物的通用名藥物(學名藥)販售。[1]
醫療用途
[编辑]利巴韋林主要用於治療慢性C型肝炎以及病毒性出血熱(在大多數國家,後者被視為孤兒藥(罕見疾病)適應症)。[10]由於美國食品藥物管理局(FDA)已發出黑框警告,禁止將其作為慢性C型肝炎的單一療法,表示此藥物在這些用途上的療效已受質疑。因此它在美國僅能作為一種或多種其他藥物的輔助療法使用。目前尚未能定論利巴韋林對於除C型肝炎病毒以外病毒(包括引起病毒性出血熱的病毒)的確切療效。事實上,美國除核准治療C型肝炎以外,並未准其用於治療其他病毒感染。[6][5]
C 型肝炎
[编辑]利巴韋林(膠囊或片劑)僅能與長效型干擾素干擾素α併用,以治療慢性C型肝炎感染。[5][11][12][13][14]他汀類藥物可能會提高此組合治療C型肝炎的療效。[15]在可能的情況下會先對特定的病毒株進行基因型鑑定。利巴韋林僅作為特定基因型與其他藥物組合中的"劑量遞增式"[a]輔助藥物。[16]
由於許多感染最終會在不經治療的情況下自行痊癒,因此對於急性C型肝炎感染(感染後的前6個月內)通常不需立即治療。[17]當決定治療急性C型肝炎時,利巴韋林可作為數種藥物組合的輔助療法 。[16]然而醫護人員在臨床上仍偏好優先使用其他藥物。[16][17]
病毒性出血熱
[编辑]利巴韋林是目前已知唯一可用於多種病毒性出血熱的治療藥物,包括拉薩熱、克里米亞–剛果出血熱、委內瑞拉出血熱以及漢他病毒感染。然而關於這些感染的數據十分稀少,且該藥物可能僅在感染初期有效。[18][19][20][21]美國陸軍傳染病醫學研究院(USAMRIID)指出:"利巴韋林在體外實驗與體內實驗中,對絲狀病毒科(伊波拉[22]與馬堡病毒)以及黃病毒屬(登革熱、黃熱病、鄂木斯克出血熱以及基阿沙努森林病)的活性均不佳" 。[23]過去曾使用噴霧形式(吸入型)來治療兒童的人類呼吸道合胞病毒相關疾病,但支持此療法的證據相當薄弱。[24]
雖然對利巴韋林的療效仍存在疑問,但它目前仍是已知唯一能有效治療拉薩熱的抗病毒藥物 。[25]
此外,利巴韋林也曾與氯胺酮 、咪達唑侖及金剛烷胺併用,嘗試用於治療狂犬病 。[26]
實驗性用途
[编辑]實驗數據顯示利巴韋林可能對犬瘟熱及痘病毒科具臨床價值。[27][28]利巴韋林也曾被用於治療單純疱疹病毒感染。一項小型研究發現使用利巴韋林治療,比安慰劑能減輕疱疹疫情爆發的嚴重程度並促進康復 。[29]另一項研究則發現利巴韋林能增強阿昔洛韋的抗病毒效果 。[30]
目前已有研究關注其在治療真核起始因子4E(eIF4E)轉譯水平升高的癌症方面的潛力,特別是急性骨髓性白血病(AML)以及頭頸癌。[31][32][33][34]在單一療法與併用療法的研究中,利巴韋林能針對AML患者體內的eIF4E產生作用,並與客觀的臨床反應(包括完全緩解)相對應。[35][36][37]然而AML患者會產生利巴韋林抗藥性,導致eIF4E標靶作用失效並造成復發。抗藥性產生的原因是由於AML細胞透過葡糖苷酸化反應,使利巴韋林失活,或是藥物進入與停留在AML細胞內的機制受損所致。[38]癌細胞可能還表現出其他形式的利巴韋林抗藥性。在與人類乳突病毒相關的口咽癌中,利巴韋林降低部分患者體內磷酸化形式的eIF4E水平。[34]在此類研究中,最佳的反應為病情穩定,但在另一項針對頭頸部的研究中則取得更有前景的結果。[33]
不良反應
[编辑]此藥物具有兩項FDA的黑框警告:其一指出,不論男女,在懷孕前或懷孕期間使用都可能導致嬰兒先天性缺陷,其二則是關於紅血球分解(溶血)的風險。[39]
利巴韋林不應與齊多夫定併用,因為會增加貧血的風險。[40]同樣地,應避免與地達諾新同時使用,因為會增加線粒體毒性的風險。[41]
作用機制
[编辑]利巴韋林是一種鳥苷(核糖核酸)類似物,用於阻斷病毒的RNA合成與病毒mRNA的加帽(指在mRNA的5'端加上一個特殊結構)過程,因此,它屬於一種核苷類似物。利巴韋林是一種前體藥物,經代謝後其結構與嘌呤RNA核苷酸相似。它在此形式下會干擾病毒複製所需的RNA代謝。目前已針對其作用機制提出超過五種直接與間接的機制假設。[42]在體外實驗中,肌苷三磷酸焦磷酸酶(ITPase)會將利巴韋林三磷酸去磷酸化為利巴韋林單磷酸(因而降低藥物活性)。而在約30%的人類體內存在的ITPase活性降低現象,能增強藥物誘導C型肝炎病毒產生突變的效果,進而提升治療成效。[43]
RNA病毒
[编辑]利巴韋林的醯胺基會因其旋轉角度,使該天然核苷藥物模擬腺苷或是鳥苷。因此,當利巴韋林作為腺嘌呤或鳥嘌呤的鹼基類似物被併入RNA時,它能與尿嘧啶或胞嘧啶同樣良好地配對,進而誘導RNA病毒在RNA依賴性複製過程中產生突變。這種高度突變對RNA病毒而言可能是致命的。[44][45]
DNA病毒
[编辑]上述機制皆無法解釋利巴韋林對多種DNA病毒的效果,目前的機制尚未被完全了解,尤其是考量到利巴韋林的2'去氧核糖類似物完全不具活性,顯示該藥物僅作為RNA核苷模擬物發揮作用,而非DNA核苷模擬物。利巴韋林5'-單磷酸會抑制細胞內的肌苷單磷酸去氫酶(IMPDH),進而耗盡細胞內的鳥苷三磷酸(GTP)庫。[46][與來源不符]
癌症中的eIF4E標靶作用
[编辑]真核起始因子4E在RNA代謝中扮演多重角色,其中以轉譯作用的研究最為詳盡。生物物理與核磁共振研究首次揭示利巴韋林能直接結合eIF4E,為其作用提供另一種機制。[47][48][49][38]在細胞實驗中,標記氚的利巴韋林(3H Ribavirin)也會與eIF4E產生交互作用。[38][32]雖然肌苷單磷酸去氫酶 (IMPDH")推測僅能與利巴韋林的單磷酸代謝物(RMP)結合,但eIF4E可結合利巴韋林,且對利巴韋林的磷酸化形式具有更高的結合親和力。[47][48][32]在許多細胞株中,針對代謝物穩態濃度的研究顯示,利巴韋林三磷酸(RTP)的含量比作為IMPDH配體的RMP代謝物更為豐富。[50][51]根據核磁共振觀察,RTP會結合在eIF4E的帽結合位點 。[49]利巴韋林會抑制細胞內的eIF4E活性,包括其在RNA輸出、轉譯以及致癌活性方面的功能 。[47][48][52][53][38][54][55][56][57][58][59][60][61]在接受利巴韋林治療的AML患者中,利巴韋林透過干擾eIF4E與其核輸入蛋白Importin 8的交互作用,阻斷eIF4E進入細胞核,進而損害其核內活性。[62][37][36][38]AML患者的臨床復發與利巴韋林結合失效有關,導致eIF4E重新進入細胞核,並恢復其核內活性。[62][37][36][38]
歷史與文化
[编辑]利巴韋林於1972年在美國國家癌症研究所(National Cancer Institute)的"病毒–癌症計畫"(Virus-Cancer program)框架下首次合成。[63]該研發工作由 Bausch Health(當時稱為國際化學與核子公司(International Chemical and Nuclear Corporation))的研究人員Roberts A. Smith、Joseph T. Witkovski與Roland K. Robins共同完成。[64]早期研究指出,利巴韋林在細胞培養與動物實驗中對多種RNA及DNA病毒均展現出抗病毒活性,且在癌症化療的背景下未產生過度毒性。[65]然而,隨著"病毒–癌症計畫"在1970年代末期被廣泛視為失敗,藥物的研發工作也一度遭到擱置。[66]
美國政府於1984年宣佈愛滋病是由反轉錄病毒所引起,科學界隨即重新檢視"病毒–癌症計畫"期間研發的藥物及其對反轉錄病毒的療效。雖然FDA於1986年首度核准利巴韋林作為抗病毒藥物,但其適應症並不包含愛滋病。這導致當時許多愛滋病患者轉而透過"買家俱樂部"(buyer's clubs)從黑市管道取得藥物。利巴韋林於1993年在美國獲准用於漢他病毒的試驗性治療,但一項非隨機且無對照組的試驗結果令人失望:受試者中高達71%出現貧血症狀,死亡率則達47%。
名稱
[编辑]此藥物的國際非專有藥名(INN)及美國採用名稱(USAN)均為Ribavirin,而在英國核准名稱則為tribavirin。市場上的學名藥商品名有Copegus、Ribasphere與Rebetol等。[1]
塔里巴韋林
[编辑]迄今為止最成功的利巴韋林衍生物,是將原有的3-甲醯胺(3-carboxamide)轉化為3-甲醯胺基 (3-carboxamidine)的衍生物,此衍生物由研究人員J. T. Witkowski等人於1973年首次提出報告,[67]現稱為塔里巴韋林(舊稱為"viramidine"或"ribamidine")。此藥物顯示出與利巴韋林相似的抗病毒譜系,它已知是利巴韋林的前體藥物。然而塔里巴韋林較利巴韋林具有更少紅血球蓄積以及更佳肝臟標靶性的優點。第一項特性歸因於塔里巴韋林的鹼性脒基(amidine group)會抑制藥物進入紅血球。而第二項特性則可能歸因於肝臟組織中將脒基轉化為醯胺基的酵素濃度較高。[68]塔里巴韋林已於2012年完成第三期人體臨床試驗。[69]
註記
[编辑]- ^ Begins with a low dose, at or near the lowest point of the expected therapeutic index, and then the dose is progressively increased until achieving desired effects.
參考文獻
[编辑]- ^ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 Ribavirin. The American Society of Health-System Pharmacists. [2016-12-08]. (原始内容存档于2016-12-20).
- ^ Ribavirin (Ibavyr). Catie. 2022 [2022-08-22].
- ^ 3.0 3.1 3.2 3.3 3.4 PRODUCT INFORMATION REBETOL (RIBAVIRIN) CAPSULES (PDF). TGA eBusiness Services. Merck Sharp & Dohme (Australia) Pty Limited. 2013-04-29 [2014-02-23]. (原始内容存档于2017-08-16).
- ^ Anvisa. RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control]. Diário Oficial da União. 2023-03-31 (2023-04-04) [2023-08-16]. (原始内容存档于2023-08-03) (巴西葡萄牙语).
- ^ 5.0 5.1 5.2 Safrin S. Chapter 49: Antiviral Agents. Katzung's Basic & Clinical Pharmacology 16th. McGraw Hill. 2024. Other antiviral agents: Ribavirin.
- ^ 6.0 6.1 Copegus: Package Insert. Drugs.com. 2023-03-27 [2024-01-19]. (原始内容存档于2025-08-07) (英语).
- ^ World Health Organization. Stuart MC, Kouimtzi M, Hill SR , 编. WHO Model Formulary 2008. World Health Organization. 2009: 177. ISBN 978-92-4-154765-9. hdl:10665/44053
 .
.
- ^ Fischer J, Ganellin CR. Analogue-based Drug Discovery. John Wiley & Sons. 2006: 504. ISBN 978-3-527-60749-5. (原始内容存档于2016-12-20).
- ^ World Health Organization. World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. hdl:10665/325771
 . WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Rebetol, Ribasphere (ribavirin) dosing, indications, interactions, adverse effects, and more. Medscape Reference. WebMD. [2014-02-23]. (原始内容存档于2014-02-28).
- ^ Paeshuyse J, Dallmeier K, Neyts J. Ribavirin for the treatment of chronic hepatitis C virus infection: a review of the proposed mechanisms of action. Current Opinion in Virology. December 2011, 1 (6): 590–598. PMID 22440916. doi:10.1016/j.coviro.2011.10.030.
- ^ Flori N, Funakoshi N, Duny Y, Valats JC, Bismuth M, Christophorou D, Daurès JP, Blanc P. Pegylated interferon-α2a and ribavirin versus pegylated interferon-α2b and ribavirin in chronic hepatitis C: a meta-analysis. Drugs. March 2013, 73 (3): 263–277. PMID 23436591. S2CID 46334977. doi:10.1007/s40265-013-0027-1.
- ^ Druyts E, Thorlund K, Wu P, Kanters S, Yaya S, Cooper CL, Mills EJ. Efficacy and safety of pegylated interferon alfa-2a or alfa-2b plus ribavirin for the treatment of chronic hepatitis C in children and adolescents: a systematic review and meta-analysis. Clinical Infectious Diseases. April 2013, 56 (7): 961–967. PMID 23243171. doi:10.1093/cid/cis1031
 .
.
- ^ Zeuzem S, Poordad F. Pegylated-interferon plus ribavirin therapy in the treatment of CHC: individualization of treatment duration according to on-treatment virologic response. Current Medical Research and Opinion. July 2010, 26 (7): 1733–1743. PMID 20482242. S2CID 206965239. doi:10.1185/03007995.2010.487038.
- ^ Zhu Q, Li N, Han Q, Zhang P, Yang C, Zeng X, Chen Y, Lv Y, Liu X, Liu Z. Statin therapy improves response to interferon alfa and ribavirin in chronic hepatitis C: a systematic review and meta-analysis. Antiviral Research. June 2013, 98 (3): 373–379. PMID 23603497. doi:10.1016/j.antiviral.2013.04.009.
- ^ 16.0 16.1 16.2 Chung RT, Ghany MG, Kim AY, Marks KM, Naggie S, Vargas HE, Aronsohn AI, Bhattacharya D, Broder T, Falade-Nwulia OO, Fontana RJ, Gordon SC, Heller T, Holmberg SD, Jhaveri R, Jonas MM, Kiser JJ, Linas BP, Lo Re V, Morgan TR, Nahass RG, Peters MG, Reddy KR, Reynolds A, Scott JD, Searson G, Swan T, Terrault NA, Trooskin SB, Wong JB. Hepatitis C Guidance 2018 Update: AASLD-IDSA Recommendations for Testing, Managing, and Treating Hepatitis C Virus Infection. Clinical Infectious Diseases. October 2018, 67 (10): 1477–1492. PMC 7190892
 . PMID 30215672. doi:10.1093/cid/ciy585. 已忽略未知参数
. PMID 30215672. doi:10.1093/cid/ciy585. 已忽略未知参数|collaboration=(帮助) - ^ 17.0 17.1 Pawlotsky J, Negro F, Aghemo A, Berenguer M, Dalgard O, Dusheiko G, Marra F, Puoti M, Wedemeyer H. EASL Recommendations on Treatment of Hepatitis C 2018. Journal of Hepatology. August 2018, 69 (2): 461–511. PMID 29650333. doi:10.1016/j.jhep.2018.03.026.
- ^ Steckbriefe seltener und importierter Infektionskrankheiten [Characteristics of rare and imported infectious diseases] (PDF). Berlin: Robert Koch Institute. 2006. ISBN 978-3-89606-095-2. (原始内容 (PDF)存档于2011-09-29).
- ^ Ascioglu S, Leblebicioglu H, Vahaboglu H, Chan KA. Ribavirin for patients with Crimean-Congo haemorrhagic fever: a systematic review and meta-analysis. The Journal of Antimicrobial Chemotherapy. June 2011, 66 (6): 1215–1222. PMID 21482564. doi:10.1093/jac/dkr136
 .
.
- ^ Bausch DG, Hadi CM, Khan SH, Lertora JJ. Review of the literature and proposed guidelines for the use of oral ribavirin as postexposure prophylaxis for Lassa fever. Clinical Infectious Diseases. December 2010, 51 (12): 1435–1441. PMC 7107935
 . PMID 21058912. doi:10.1086/657315.
. PMID 21058912. doi:10.1086/657315.
- ^ Soares-Weiser K, Thomas S, Thomson G, Garner P. Ribavirin for Crimean-Congo hemorrhagic fever: systematic review and meta-analysis. BMC Infectious Diseases. July 2010, 10. PMC 2912908
 . PMID 20626907. doi:10.1186/1471-2334-10-207
. PMID 20626907. doi:10.1186/1471-2334-10-207  . 已忽略未知参数
. 已忽略未知参数|article-number=(帮助) - ^ Goeijenbier M, van Kampen JJ, Reusken CB, Koopmans MP, van Gorp EC. Ebola virus disease: a review on epidemiology, symptoms, treatment and pathogenesis. The Netherlands Journal of Medicine. November 2014, 72 (9): 442–448. PMID 25387613. (原始内容存档于2014-11-29).
- ^ Medical Management of Biological Casualties Handbook. United States Government Printing Office. 2011: 115. ISBN 978-0-16-090015-0.
- ^ Ventre K, Randolph AG. Ventre K , 编. Ribavirin for respiratory syncytial virus infection of the lower respiratory tract in infants and young children. The Cochrane Database of Systematic Reviews. January 2007, (1). PMID 17253446. doi:10.1002/14651858.CD000181.pub3. 已忽略未知参数
|article-number=(帮助) - ^ Treatment | Lassa Fever | CDC. 2023-09-26 [2024-01-20]. (原始内容存档于2023-09-26).
- ^ Hemachudha T, Ugolini G, Wacharapluesadee S, Sungkarat W, Shuangshoti S, Laothamatas J. Human rabies: neuropathogenesis, diagnosis, and management. The Lancet. Neurology. May 2013, 12 (5): 498–513. PMID 23602163. S2CID 1798889. doi:10.1016/S1474-4422(13)70038-3.
- ^ Elia G, Belloli C, Cirone F, Lucente MS, Caruso M, Martella V, Decaro N, Buonavoglia C, Ormas P. In vitro efficacy of ribavirin against canine distemper virus. Antiviral Research. February 2008, 77 (2): 108–113. PMID 17949825. doi:10.1016/j.antiviral.2007.09.004.
- ^ Baker RO, Bray M, Huggins JW. Potential antiviral therapeutics for smallpox, monkeypox and other orthopoxvirus infections. Antiviral Research. January 2003, 57 (1–2): 13–23. PMC 9533837
 . PMID 12615299. doi:10.1016/S0166-3542(02)00196-1.
. PMID 12615299. doi:10.1016/S0166-3542(02)00196-1.
- ^ Bierman SM, Kirkpatrick W, Fernandez H. Clinical efficacy of ribavirin in the treatment of genital herpes simplex virus infection. Chemotherapy. 1981, 27 (2): 139–145. PMID 7009087. doi:10.1159/000237969.
- ^ Pancheva SN. Potentiating effect of ribavirin on the anti-herpes activity of acyclovir. Antiviral Research. September 1991, 16 (2): 151–161. PMID 1665959. doi:10.1016/0166-3542(91)90021-I.
- ^ Kast RE. Ribavirin in cancer immunotherapies: controlling nitric oxide helps generate cytotoxic lymphocyte. Cancer Biology & Therapy. November–December 2002, 1 (6): 626–630. PMID 12642684. S2CID 24010404. doi:10.4161/cbt.310
 .
.
- ^ 32.0 32.1 32.2 Borden KL, Culjkovic-Kraljacic B. Ribavirin as an anti-cancer therapy: acute myeloid leukemia and beyond?. Leukemia & Lymphoma. October 2010, 51 (10): 1805–1815. PMC 2950216
 . PMID 20629523. doi:10.3109/10428194.2010.496506.
. PMID 20629523. doi:10.3109/10428194.2010.496506.
- ^ 33.0 33.1 Dunn LA, Fury MG, Sherman EJ, Ho AA, Katabi N, Haque SS, Pfister DG. Phase I study of induction chemotherapy with afatinib, ribavirin, and weekly carboplatin and paclitaxel for stage IVA/IVB human papillomavirus-associated oropharyngeal squamous cell cancer. Head & Neck. February 2018, 40 (2): 233–241. PMC 6760238
 . PMID 28963790. doi:10.1002/hed.24938.
. PMID 28963790. doi:10.1002/hed.24938.
- ^ 34.0 34.1 Burman B, Drutman SB, Fury MG, Wong RJ, Katabi N, Ho AL, Pfister DG. Pharmacodynamic and therapeutic pilot studies of single-agent ribavirin in patients with human papillomavirus-related malignancies. Oral Oncology. May 2022, 128. PMC 9788648
 . PMID 35339025. doi:10.1016/j.oraloncology.2022.105806. 已忽略未知参数
. PMID 35339025. doi:10.1016/j.oraloncology.2022.105806. 已忽略未知参数|article-number=(帮助) - ^ Assouline S, Culjkovic B, Cocolakis E, Rousseau C, Beslu N, Amri A, Caplan S, Leber B, Roy DC, Miller WH, Borden KL. Molecular targeting of the oncogene eIF4E in acute myeloid leukemia (AML): a proof-of-principle clinical trial with ribavirin. Blood. July 2009, 114 (2): 257–260. PMID 19433856. S2CID 28957125. doi:10.1182/blood-2009-02-205153
 .
.
- ^ 36.0 36.1 36.2 Assouline S, Culjkovic-Kraljacic B, Bergeron J, Caplan S, Cocolakis E, Lambert C, Lau CJ, Zahreddine HA, Miller WH, Borden KL. A phase I trial of ribavirin and low-dose cytarabine for the treatment of relapsed and refractory acute myeloid leukemia with elevated eIF4E. Haematologica. January 2015, 100 (1): e7–e9. PMC 4281321
 . PMID 25425688. doi:10.3324/haematol.2014.111245.
. PMID 25425688. doi:10.3324/haematol.2014.111245.
- ^ 37.0 37.1 37.2 Assouline S, Gasiorek J, Bergeron J, Lambert C, Culjkovic-Kraljacic B, Cocolakis E, Zakaria C, Szlachtycz D, Yee K, Borden KL. Molecular targeting of the UDP-glucuronosyltransferase enzymes in high-eukaryotic translation initiation factor 4E refractory/relapsed acute myeloid leukemia patients: a randomized phase II trial of vismodegib, ribavirin with or without decitabine. Haematologica. March 2023, 108 (11): 2946–2958. PMC 10620574
 . PMID 36951168. S2CID 257733013. doi:10.3324/haematol.2023.282791
. PMID 36951168. S2CID 257733013. doi:10.3324/haematol.2023.282791  .
.
- ^ 38.0 38.1 38.2 38.3 38.4 38.5 Zahreddine HA, Culjkovic-Kraljacic B, Assouline S, Gendron P, Romeo AA, Morris SJ, Cormack G, Jaquith JB, Cerchietti L, Cocolakis E, Amri A, Bergeron J, Leber B, Becker MW, Pei S, Jordan CT, Miller WH, Borden KL. The sonic hedgehog factor GLI1 imparts drug resistance through inducible glucuronidation. Nature. July 2014, 511 (7507): 90–93. Bibcode:2014Natur.511...90Z. PMC 4138053
 . PMID 24870236. doi:10.1038/nature13283.
. PMID 24870236. doi:10.1038/nature13283.
- ^ Copedgus (PDF). FDA.gov. [16 April 2017]. (原始内容存档 (PDF)于2014-11-03).
- ^ Alvarez D, Dieterich DT, Brau N, Moorehead L, Ball L, Sulkowski MS. Zidovudine use but not weight-based ribavirin dosing impacts anaemia during HCV treatment in HIV-infected persons. Journal of Viral Hepatitis. October 2006, 13 (10): 683–689. PMID 16970600. S2CID 21474337. doi:10.1111/j.1365-2893.2006.00749.x.
- ^ Bani-Sadr F, Carrat F, Pol S, Hor R, Rosenthal E, Goujard C, Morand P, Lunel-Fabiani F, Salmon-Ceron D, Piroth L, Pialoux G, Bentata M, Cacoub P, Perronne C. Risk factors for symptomatic mitochondrial toxicity in HIV/hepatitis C virus-coinfected patients during interferon plus ribavirin-based therapy. Journal of Acquired Immune Deficiency Syndromes. September 2005, 40 (1): 47–52. PMID 16123681. S2CID 9364536. doi:10.1097/01.qai.0000174649.51084.46
 . 已忽略未知参数
. 已忽略未知参数|collaboration=(帮助) - ^ Graci JD, Cameron CE. Mechanisms of action of ribavirin against distinct viruses. Reviews in Medical Virology. January 2006, 16 (1): 37–48. PMC 7169142
 . PMID 16287208. doi:10.1002/rmv.483.
. PMID 16287208. doi:10.1002/rmv.483.
- ^ Nyström K, Wanrooij PH, Waldenström J, Adamek L, Brunet S, Said J, Nilsson S, Wind-Rotolo M, Hellstrand K, Norder H, Tang KW, Lagging M. Inosine Triphosphate Pyrophosphatase Dephosphorylates Ribavirin Triphosphate and Reduced Enzymatic Activity Potentiates Mutagenesis in Hepatitis C Virus. Journal of Virology. October 2018, 92 (19): 01087–18. PMC 6146798
 . PMID 30045981. doi:10.1128/JVI.01087-18
. PMID 30045981. doi:10.1128/JVI.01087-18  . 已忽略未知参数
. 已忽略未知参数|article-number=(帮助) - ^ Ortega-Prieto AM, Sheldon J, Grande-Pérez A, Tejero H, Gregori J, Quer J, Esteban JI, Domingo E, Perales C. Vartanian JP , 编. Extinction of hepatitis C virus by ribavirin in hepatoma cells involves lethal mutagenesis. PLOS ONE. 2013, 8 (8). Bibcode:2013PLoSO...871039O. PMC 3745404
 . PMID 23976977. doi:10.1371/journal.pone.0071039
. PMID 23976977. doi:10.1371/journal.pone.0071039  . 已忽略未知参数
. 已忽略未知参数|article-number=(帮助) - ^ Crotty S, Cameron C, Andino R. Ribavirin's antiviral mechanism of action: lethal mutagenesis?. Journal of Molecular Medicine. February 2002, 80 (2): 86–95. PMID 11907645. S2CID 52851826. doi:10.1007/s00109-001-0308-0.
- ^ Leyssen P, De Clercq E, Neyts J. The anti-yellow fever virus activity of ribavirin is independent of error-prone replication. Molecular Pharmacology. April 2006, 69 (4): 1461–1467. PMID 16421290. S2CID 8158590. doi:10.1124/mol.105.020057.
- ^ 47.0 47.1 47.2 Kentsis A, Topisirovic I, Culjkovic B, Shao L, Borden KL. Ribavirin suppresses eIF4E-mediated oncogenic transformation by physical mimicry of the 7-methyl guanosine mRNA cap. Proceedings of the National Academy of Sciences of the United States of America. December 2004, 101 (52): 18105–18110. Bibcode:2004PNAS..10118105K. PMC 539790
 . PMID 15601771. doi:10.1073/pnas.0406927102
. PMID 15601771. doi:10.1073/pnas.0406927102  .
.
- ^ 48.0 48.1 48.2 Kentsis A, Volpon L, Topisirovic I, Soll CE, Culjkovic B, Shao L, Borden KL. Further evidence that ribavirin interacts with eIF4E. RNA. December 2005, 11 (12): 1762–1766. PMC 1370864
 . PMID 16251386. doi:10.1261/rna.2238705.
. PMID 16251386. doi:10.1261/rna.2238705.
- ^ 49.0 49.1 Volpon L, Osborne MJ, Zahreddine H, Romeo AA, Borden KL. Conformational changes induced in the eukaryotic translation initiation factor eIF4E by a clinically relevant inhibitor, ribavirin triphosphate. Biochemical and Biophysical Research Communications. May 2013, 434 (3): 614–619. Bibcode:2013BBRC..434..614V. PMC 3659399
 . PMID 23583375. doi:10.1016/j.bbrc.2013.03.125.
. PMID 23583375. doi:10.1016/j.bbrc.2013.03.125.
- ^ Smee DF, Matthews TR. Metabolism of ribavirin in respiratory syncytial virus-infected and uninfected cells. Antimicrobial Agents and Chemotherapy. July 1986, 30 (1): 117–121. PMC 176447
 . PMID 3752974. doi:10.1128/AAC.30.1.117.
. PMID 3752974. doi:10.1128/AAC.30.1.117.
- ^ Page T, Connor JD. The metabolism of ribavirin in erythrocytes and nucleated cells. The International Journal of Biochemistry. January 1990, 22 (4): 379–383. PMID 2159925. doi:10.1016/0020-711X(90)90140-X.
- ^ Pettersson F, Yau C, Dobocan MC, Culjkovic-Kraljacic B, Retrouvey H, Puckett R, Flores LM, Krop IE, Rousseau C, Cocolakis E, Borden KL, Benz CC, Miller WH. Ribavirin treatment effects on breast cancers overexpressing eIF4E, a biomarker with prognostic specificity for luminal B-type breast cancer. Clinical Cancer Research. May 2011, 17 (9): 2874–2884. PMC 3086959
 . PMID 21415224. doi:10.1158/1078-0432.CCR-10-2334.
. PMID 21415224. doi:10.1158/1078-0432.CCR-10-2334.
- ^ Bollmann F, Fechir K, Nowag S, Koch K, Art J, Kleinert H, Pautz A. Human inducible nitric oxide synthase (iNOS) expression depends on chromosome region maintenance 1 (CRM1)- and eukaryotic translation initiation factor 4E (elF4E)-mediated nucleocytoplasmic mRNA transport. Nitric Oxide. April 2013, 30: 49–59. PMID 23471078. doi:10.1016/j.niox.2013.02.083.
- ^ Shi F, Len Y, Gong Y, Shi R, Yang X, Naren D, Yan T. Eaves CJ , 编. Ribavirin Inhibits the Activity of mTOR/eIF4E, ERK/Mnk1/eIF4E Signaling Pathway and Synergizes with Tyrosine Kinase Inhibitor Imatinib to Impair Bcr-Abl Mediated Proliferation and Apoptosis in Ph+ Leukemia. PLOS ONE. 2015-08-28, 10 (8). Bibcode:2015PLoSO..1036746S. PMC 4552648
 . PMID 26317515. doi:10.1371/journal.pone.0136746
. PMID 26317515. doi:10.1371/journal.pone.0136746  . 已忽略未知参数
. 已忽略未知参数|article-number=(帮助) - ^ Zismanov V, Attar-Schneider O, Lishner M, Heffez Aizenfeld R, Tartakover Matalon S, Drucker L. Multiple myeloma proteostasis can be targeted via translation initiation factor eIF4E. International Journal of Oncology. February 2015, 46 (2): 860–870. PMID 25422161. doi:10.3892/ijo.2014.2774
 .
.
- ^ Dai D, Chen H, Tang J, Tang Y. Inhibition of mTOR/eIF4E by anti-viral drug ribavirin effectively enhances the effects of paclitaxel in oral tongue squamous cell carcinoma. Biochemical and Biophysical Research Communications. January 2017, 482 (4): 1259–1264. Bibcode:2017BBRC..482.1259D. PMID 27932243. doi:10.1016/j.bbrc.2016.12.025.
- ^ Volpin F, Casaos J, Sesen J, Mangraviti A, Choi J, Gorelick N, Frikeche J, Lott T, Felder R, Scotland SJ, Eisinger-Mathason TS, Brem H, Tyler B, Skuli N. Use of an anti-viral drug, Ribavirin, as an anti-glioblastoma therapeutic. Oncogene. May 2017, 36 (21): 3037–3047. PMID 27941882. S2CID 21655102. doi:10.1038/onc.2016.457.
- ^ Wang G, Li Z, Li Z, Huang Y, Mao X, Xu C, Cui S. Targeting eIF4E inhibits growth, survival and angiogenesis in retinoblastoma and enhances efficacy of chemotherapy. Biomedicine & Pharmacotherapy. December 2017, 96: 750–756. PMID 29049978. doi:10.1016/j.biopha.2017.10.034.
- ^ Xi C, Wang L, Yu J, Ye H, Cao L, Gong Z. Inhibition of eukaryotic translation initiation factor 4E is effective against chemo-resistance in colon and cervical cancer. Biochemical and Biophysical Research Communications. September 2018, 503 (4): 2286–2292. Bibcode:2018BBRC..503.2286X. PMID 29959920. S2CID 49634908. doi:10.1016/j.bbrc.2018.06.150.
- ^ Jin J, Xiang W, Wu S, Wang M, Xiao M, Deng A. Targeting eIF4E signaling with ribavirin as a sensitizing strategy for ovarian cancer. Biochemical and Biophysical Research Communications. March 2019, 510 (4): 580–586. Bibcode:2019BBRC..510..580J. PMID 30739792. S2CID 73419809. doi:10.1016/j.bbrc.2019.01.117.
- ^ Urtishak KA, Wang LS, Culjkovic-Kraljacic B, Davenport JW, Porazzi P, Vincent TL, Teachey DT, Tasian SK, Moore JS, Seif AE, Jin S, Barrett JS, Robinson BW, Chen IL, Harvey RC, Carroll MP, Carroll AJ, Heerema NA, Devidas M, Dreyer ZE, Hilden JM, Hunger SP, Willman CL, Borden KL, Felix CA. Targeting EIF4E signaling with ribavirin in infant acute lymphoblastic leukemia. Oncogene. March 2019, 38 (13): 2241–2262. PMC 6440839
 . PMID 30478448. doi:10.1038/s41388-018-0567-7.
. PMID 30478448. doi:10.1038/s41388-018-0567-7.
- ^ 62.0 62.1 Volpon L, Culjkovic-Kraljacic B, Osborne MJ, Ramteke A, Sun Q, Niesman A, Chook YM, Borden KL. Importin 8 mediates m7G cap-sensitive nuclear import of the eukaryotic translation initiation factor eIF4E. Proceedings of the National Academy of Sciences of the United States of America. May 2016, 113 (19): 5263–5268. Bibcode:2016PNAS..113.5263V. PMC 4868427
 . PMID 27114554. doi:10.1073/pnas.1524291113
. PMID 27114554. doi:10.1073/pnas.1524291113  .
.
- ^ Snell NJ. Ribavirin--current status of a broad spectrum antiviral agent. Expert Opinion on Pharmacotherapy. August 2001, 2 (8): 1317–1324. PMID 11585000. S2CID 46564870. doi:10.1517/14656566.2.8.1317.
- ^ Ribavirin History. News-Medical.net. 2010-04-12 [2016-02-19]. (原始内容存档于2016-03-02).
- ^ Sidwell RW, Huffman JH, Khare GP, Allen LB, Witkowski JT, Robins RK. Broad-spectrum antiviral activity of Virazole: 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide. Science. August 1972, 177 (4050): 705–706. Bibcode:1972Sci...177..705S. PMID 4340949. S2CID 43106875. doi:10.1126/science.177.4050.705.
- ^ Sneader, Walter. Drug Discovery: A History. Chichester: John Wiley & Sons. 2005: 262. ISBN 978-0-471-89980-8.
- ^ Witkowski JT, Robins RK, Khare GP, Sidwell RW. Synthesis and antiviral activity of 1,2,4-triazole-3-thiocarboxamide and 1,2,4-triazole-3-carboxamidine ribonucleosides. Journal of Medicinal Chemistry. August 1973, 16 (8): 935–937. PMID 4355593. doi:10.1021/jm00266a014.
- ^ Sidwell RW, Bailey KW, Wong MH, Barnard DL, Smee DF. In vitro and in vivo influenza virus-inhibitory effects of viramidine. Antiviral Research. October 2005, 68 (1): 10–17. PMID 16087250. doi:10.1016/j.antiviral.2005.06.003.
- ^ U.S. National Library of Medicine. Study of Viramidine to Ribavirin in Patients With Chronic Hepatitis C Who Are Treatment Naive (VISER2) (报告). National Institutes of Health. 2012-06-22 [2021-03-25]. (原始内容存档于2023-06-21).
