Upjohn
外觀
| The Upjohn Company | |
|---|---|
 | |
| 公司類型 | 公司 |
| 公司結局 | 與Pharmacia合併為Pharmacia & Upjohn |
| 後繼機構 | Pharmacia & Upjohn 暉致(Viatris) |
| 成立 | 1886年 |
| 結束 | 1995年 |
| 總部 | 美國密歇根州波蒂奇 |
| 產業 | 製藥產業 |

Upjohn是一家美國製藥公司,1886年由威廉·E·厄普約翰在密歇根州黑斯廷斯成立[1],1995年與瑞典Pharmacia公司合併為Pharmacia & Upjohn,現為輝瑞公司所有。
歷史
[編輯]1976年,公司的三位科學家開發了Upjohn雙羥基化反應[2]。
產品
[編輯]四位數
[編輯]| 名稱 | CAS號 | 分子式 | 化學結構 |
|---|---|---|---|
| 苄非他明 (U-0441) |
156-08-1 | C 17H 21N |

|
| U-0882 | 310-35-0 | C 18H 21NO 2 |

|
| 敵鼠 (U-1363) |
82-66-6 | C 23H 16O 3 |

|
| 羧雌醚 (U-2911) |
1755-52-8 | C 17H 22O 3 |

|
| U-4527 | 66-81-9 | C 15H 23NO 4 |

|
| U-5956 | 480-49-9 | C 35H 58O 11 |

|
| U-6040 | 76-43-7 | C 20H 29FO 3 |

|
| U-6056 | 13117-35-6 | C 13H 18N 2 |

|
| U-8113 | 1688-71-7 | C 10H 13NO |

|
| U-8344 | 66-75-1 | C 8H 11Cl 2N 3O 2 |

|
| U-8471 | 2668-66-8 | C 22H 32O 3 |

|
| U-8614 | 426-13-1 | C 22H 29FO 4 |

|
| 醋酸甲羥孕酮 (U-8839) |
71-58-9 | C 24H 34O 4 |

|
| U-8840 | 520-85-4 | C 22H 32O 3 |

|
| 2-氨基-1,1,3-三氰基-1-丙烯 (U-9189) |
868-54-2 | C 6H 4N 4 |

|
| U-9889 | 18883-66-4 | C 8H 15N 3O 7 |

|
五位數
[編輯]六位數
[編輯]| 名稱 | CAS號 | 分子式 | 化學結構 |
|---|---|---|---|
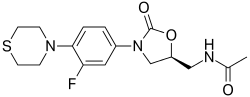
| U-100480 | 168828-58-8 | C 16H 20FN 3O 3S |
|
| U-101387 | 170858-33-0 | C 21H 27N 3O 3S |

|
| U-101958 | 170856-57-2 | C 21H 29N 3O |

|
| U-103017 | 166335-18-8 | C 28H 28N 2O 5S |
參考文獻
[編輯]- ^ Lohrstorfer, Martha; Larson, Catherine. William E. Upjohn: Person of the Century 1853 - 1932. Kalamazoo Public Library. 2002 [December 24, 2024]. (原始內容存檔於May 17, 2008).
Known by his contemporaries as a dreamer and a tinkerer, Dr. Upjohn saw a need to improve the means of administering medicine. Most medicines of the day were in fluid form, and those in pill form were often hard and insoluble. Patients were left to try to digest the bitter medicine, with no guarantee that it would dissolve in their systems effectively. Dr. Upjohn began experimenting with making better pills in the attic of his home. Eventually he invented his "friable" pill. Friable meant that the pill could easily be crushed to a powder. The pill was patented in 1885, and its reputation quickly spread within the medical community, thanks greatly to Dr. Upjohn's marketing strategy. He sent small pine boards to thousands of physicians along with samples of his rival's hard pills, and his own friable pills. He invited doctors to hammer the pills into the boards to see which one would be the most digestible. This tactic was eventually modified, but for the next 60 years, a thumb reducing an Upjohn pill to powder was used as the trademark symbol of his company, the Upjohn Pill and Granule Company, later more widely known to the world as The Upjohn Company.
- ^ VanRheenen, V.; Kelly, R.C.; Cha, D.Y. An improved catalytic OsO4 oxidation of olefins to -1,2-glycols using tertiary amine oxides as the oxidant. Tetrahedron Letters. 1976-06, 17 (23): 1973–1976. doi:10.1016/S0040-4039(00)78093-2.
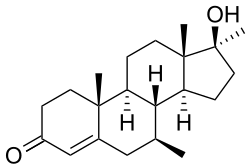
- ^ 3.0 3.1 ((Lyster, S C.)), ((Duncan, G W.)). ANABOLIC, ANDROGENIC AND MYOTROPIC ACTIVITIES OF DERIVATIVES OF 7α-METHYL-19-NORTESTOSTERONE. Acta Endocrinologica. July 1963, 43 (3): 399–411. doi:10.1530/acta.0.0430399. 溫哥華格式錯誤 (幫助)
- ^ ((Castañer, J.)), ((Hillier, K.)). U-23,469. Drugs of the Future. 1980, 5 (11): 564. ISSN 0377-8282. doi:10.1358/dof.1980.005.11.1002363.